Thermal Processing of Food 101: An introduction to Pasteurization
One of the most common ways food manufacturers preserve and ensure the safety of food is through utilizing temperatures to reduce or destroy microbial and/or enzyme activity. This is the heart of thermal processing of foods and heat processing of food.
In other instances, the application of temperatures can also be used to achieve physical or chemical changes to make food products reach a certain quality or standard.
Pasteurization and sterilization are two of the main processes for thermal processing.
The purpose of this blog is to provide a straight forward overview of Pasteurization, answering the question, how does pasteurization preserve foods? This includes its historical origins, its intended purposes, the pasteurization process itself and its effect on food quality, other novel methods of pasteurization, and ultimately the role all of which play in today’s modern food safety management systems.
History of Pasteurization
The origins of pasteurization can be traced back to 1117 AD where Chinese records indicate that wine was heated in order to preserve it.
References to similar processes were sparse until 1768 when Lazzaro Spallanzani, an Italian priest, and scientist, proved that broth could be made “sterile” after it was subjected to thermal processing.
Soon after a Parisian chef named Nicolas Appert began experimenting with ways to preserve a variety of different foods in 1795 by placing food in wax-sealed glass jars and boiling it at low temperatures.
For his efforts, Appert would go on to win an award from the French army during the height of the Napoleonic Wars in 1810 as his method allowed them to reinforce their front lines which were stretching across Europe.
With his winnings, Appert published the first cookbook on modern food preservation methods, L’Art de conserver les substances animales et végétales (or The Art of Preserving Animal and Vegetable Substances), and soon after established the first food-bottling factory in the world, in Massy, a Parisian suburb.
At the same time, Peter Durand, a British merchant, patented a method for preservation using iron cans with a thin tin lid, creating the modern-day process of canning foods, however, the can opener was not to be invented for another half-century or so.
Although both Durand and Appert knew their methods worked, they did not fully understand the reason ‘why’ and it was not until 1864 that a French chemist on holiday in the town of Arbois was so put off by the acidity of locally aged wines explored why exposing them to a mild temperature of 50–60 °C (122–140 °F) for a short time would allow the wines to age better and kill the inherent microbes.
This scientist’s name was Louis Pasteur and it is in his honor that the modern process of “pasteurization” came to be known.
Purpose
Pasteurization is a relatively mild heat treatment in which food or beverages are heated to less than 100°C to eliminate pathogens or to reduce the number of spoilage organisms in a product.
It often occurs in a continuous system and utilizes plate heat exchangers and/or hot water and steam, both directly or indirectly.
It is widely used in the food industry and frequently employed as a CCP in various HACCP Plans (Note: For more information on determining when and where best to apply CCP’s this topic was previously discussed in our blog ‘Understanding the difference between PRP, OPRP & CCP’)
In food processing it is used to achieve two primary goals:
- Food safety (to destroy enzymes and relatively heat-sensitive micro-organisms such as non-spore-forming bacteria, yeast, molds, and vegetative bacteria).
- Extend the shelf life of products from a period of days to months depending on the product and pasteurization method applied.
Food pasteurization is most commonly used for milk, bottled/canned fruit and vegetables, beer, wine, fruit, and carbonated juices and can be used to prevent diseases such as tuberculosis, diphtheria, scarlet fever, and many others. This demonstrates clearly how does pasteurization preserve foods.
The scope of the parameters used in the pasteurization process is determined mostly by the natural acidity of the food but can also consider the nutritional quality and sensory characteristics of finished treated foods as well.
The scope of parameters used in pasteurization are determined mostly by the natural acidity of the food
In high acidic foods, i.e., those with a pH of less than 4.5 such as fruit juice and beer, pasteurization is applied to destroy pathogenic bacteria and microbes, as well as inactive enzymes against spoilage.
In foods with a lower acidic value, i.e., those with a pH value of more than 4.6 such as milk and liquid eggs, pasteurization is often applied to eliminate pathogens and spoilage organisms (yeast and molds).
Bacteria killed by pasteurization include Salmonella, Listeria, Campylobacter, and Escherichia coli 0157:H7 (E.coli), however, it is important to note that as the heat exposure is often mild to preserve the sensory or nutritional qualities of the food, the parameters of pasteurization is not intended or designed to destroy all spoilage organisms and that finished pasteurized products should be utilized in conjunction with other preservation methods such as refrigeration or pH reduction.
For more information on CCPs for pathogenic bacteria, download our Microbiological Hazard Datasheet Ebook:
One other caveat to keep in mind is that pasteurization does intend to kill spore-bearing microorganisms, but rather these and most other spore-bearing bacteria cannot grow in acidic fruit juices and consequently their presence is of no practical significance. This contributes to the effectiveness of thermal processing of foods.
What this means in real effect for manufacturers of juices is that the parameters used when pasteurizing products need to only be applied for such a time and level of heat that yeasts and molds are destroyed.
Ultimately, there is a dual-incentive for companies to pasteurize their products as it makes the finished product safer for consumption and has the added benefit of extending the shelf life of the product as well.
Pasteurization provides manufacturers a dual-incentive as not only does it make the finished product safer for consumption, but it also extends its shelf life
While products that are not pasteurized can still be consumed, there is usually a financial motivation for companies to do so as it results in less waste, protection for liability, and ensures customers are consuming the safest possible version of the product.
In certain circumstances however a business may find cause for not applying pasteurization, such as craft breweries who may opt to forgo the process when the final product is not intended for mass distribution but to be consumed on-site where the storage and serving conditions can be monitored to mitigate against increased inherent risks and accelerated spoilage.
Pasteurization process
There are different options a business can consider when implementing a pasteurization process from how they apply the parameters, to the actual process itself when doing so.
Time and temperature are the required parameters for pasteurization, however, how they are applied can vary from:
- heating foods to a relatively lower temperature and maintaining it for a longer time, such as Vat/batch/holding pasteurization, or
- heating foods to a high temperature and holding it for a short time only such as flash pasteurization
Pasteurization can be performed in two ways:
- filling sterile containers with the product and then pasteurizing, or
- pasteurizing the product first and then filling in sterile containers.
The time required for the heat treatment is determined by the D-value of the most heat resistant enzyme or micro-organism which may be present in the product.
The D-value is the decimal reduction time, which is the time required to reduce the targeted microorganisms by a factor of 10 / 90% of the initial load, or one log10 unit.
The z value indicates the amount of change in temperature (°C) that is required to shift the D value by 90%, or one log10 unit.
Processing times necessary to achieve a certain D value rapidly decrease with minimal increase in temperature.
For example, an analysis of the parameters and their application for milk highlights how a moderate increase in temperature has a significant impact on the required time to destroy the most heat-sensitive of the non-spore-forming pathogenic organisms.
| Temperature | Time |
|---|---|
| 63°C (145°F) | For 30 min (Low Temperature Long Time, LTLT) |
| 72°C (162°F) | For 15 sec (High Temperature Short Time, HTST) |
| 89°C (192°F) | For 1.0 sec |
| 90°C (194°F) | For 0.5 sec |
| 94°C (201°F) | For 0.1 sec |
| 100°C (212°F) | For 0.01 sec |
| 135°C (275°F) | For 2.0 – 5.0 seconds (Ultra Heat Treatment, UHT) |
Table: Milk Pasteurizing Temperatures
While milk pasteurization temperatures are sufficient to destroy all yeasts, molds, gram-negative, and many gram-positive bacteria, some micro-organisms do survive. This gives a practical perspective on the question, how does pasteurization preserve foods.
These are:
- Thermoduric: which can survive exposure to relatively high temperatures but do not necessarily grow at these temperatures, such as Streptococcus and Lactobacillus.
- Thermophilic: which can not only survive but require high temperatures to grow.
Once the D and Z values are established and known, the pasteurization method can be chosen to produce an intended commercial or sensory result.
For example, HTST pasteurization of milk will provide a shelf life of approximately two weeks if refrigerated while preserving its natural sensory properties.
Alternatively, UHT milk is a sterilization treatment which exposes the milk to 135°C (275°F) for 2–5 seconds and although it provides a comparable level of safety when accompanied by the right packaging, it will increase the shelf life of the product significantly to approximately six to nine months.
To increase the shelf-life and mitigate against temperature abuse, UHT-treated products can also be stored in refrigerated conditions and kept in opaque containers.
It is important to note that while the commercial incentive for UHT can be strong, it can cause a Maillard reaction during production and storage, and by doing so alter the sensory qualities of the treated product.
As such, despite the commercial incentive for its use, the popularity of UHT varies from region to region.
Given that products can be assumed to be consumed quite quickly once opened, it is also not uncommon for some countries like the UK and Ireland to prefer HTST while noting that in those regions where it is applied, a wide amount of variance in approaches for how UHT products are stored.
For instance, in the US UHT products such as milk may still be refrigerated, whereas in continental Europe it may be stored at ambient temperatures.
Finally, the process for confirming the effectiveness of treatment is the same regardless of the method applied.
Alkaline phosphatase is a naturally occurring enzyme in raw milk which has a similar D value to heat-resistant pathogens and as such, its presence detected typically indicates that the pasteurization conducted has been inadequate.
Effects on nutritional and sensory quality
Pasteurization typically involves relatively mild heat treatment so even when it is combined with other unit operations like irradiation, chilling, or canning in most circumstances it has a relatively minor effect on the sensory properties of the food.
However, if a business seeks a longer self-life they may opt for UHT treatment which can invoke a Maillard reaction in the food and change the sensory properties of it.
Where a Maillard reaction is not created, exposure to high temperatures can still degrade the sensory quality of the food or reduce its nutritional value through the destruction of vitamins and protein components of the food.
As such, deciding the optimum approach to pasteurization can result in a balanced consideration between safety and taste.
Alternatively, a business may forgo pasteurization completely to preserve taste in circumstances where the environment can be controlled safely and appropriately.
In these instances, such as the example of the craft brewery provided earlier, consumption occurs on-site shortly after production with no delays or variance in storage conditions being observed that increase the risk of the product.
With specific regard to nutrition, pasteurization, or the lack thereof, can result in both positive and negative nutritional outcomes depending on what outcome is being sought.
Specifically, for milk, pasteurization causes an increase of Vitamin A concentrations but also causes a significant decrease in Vitamin B2.
In fruit juices such as orange, the Vitamin C content in raw orange juice should be in higher concentration than its pasteurized equivalent and there may be a loss in sensory quality which can be attributed to the loss of volatiles.
When such situations occur, these can be re-added or may even be recovered at a later stage to produce a higher quality product, but if producing milk, you may wish to remove the volatiles accordingly to produce a more aromatic product.
Alternatively, if wishing to forgo this process – other methods might be considered.
Alternative pasteurization methods
The processes discussed so far are arguably the most prominent and commercially viable applications of preserving food for safe consumption at the time of writing, however other methods do exist and are used.
These methods can utilize both thermal and non-thermal approaches as a means to circumvent the degradation of heat-labile nutrients and preserve the nutritional values and sensory quality of the food. Here we see alternative methods to traditional thermal processing of foods that are emerging in the industry.”
Alternative pasteurization methods seek to preserve the nutritional and sensory values of the food
Non-thermal pasteurization methods are:
- High-pressure processing (HPP) and
- Pulsed Electric Field (PEF).
New thermal pasteurization methods are:
- Microwave Volumetric Heating (MVH) and
- Low Temperature, Short Time (LTST) / Millisecond Technology (MST).
HPP is probably the most common alternative to pasteurization and exposes the packaged food to a high degree of pressure to deactivate the targeted microorganisms.
It ensures food maintains its freshness throughout the shelf life.
Other names for it include hydrostatic pressure processing, ultra-high pressure processing, Pascalization, and/or cold pasteurization.
PEF can be used in a continuous flow process and utilizes electrodes to perforate cell membranes of targeted microorganisms to destroy them.
The treatments applied are incredibly short (measured in microseconds) and it requires little energy to conduct while not affecting flavor or taste, making it an attractive proposition.
While the process and logic of its application appear to be sound and it is used by companies, its wide-spread adoption has been limited due to its economic viability when scaled to an industrial requirement.
MVH was developed by AMT for pasteurizing liquids in a continuous flow and delivers microwave energy into the center of the targeted food/beverage.
By applying energy evenly throughout the food at the same time it avoids some of the pitfalls of traditional pasteurization techniques which target the surface of the food/beverage and can require less energy to operate.
LTST/MST occurs in a process chamber where the milk, in the form of droplets, is exposed for milliseconds to a temperature equal to or less than the required temperature for pasteurization (i.e., <72°C/<162°F).
Its first commercial use was in 2019 in Puerto Rico, but studies have shown that it is effective and significantly decreases microbial load to a point that the reproduction of spoilage microorganisms can take longer.
It can also be used in conjunction with HTST and UHT to extend shelf life even further.
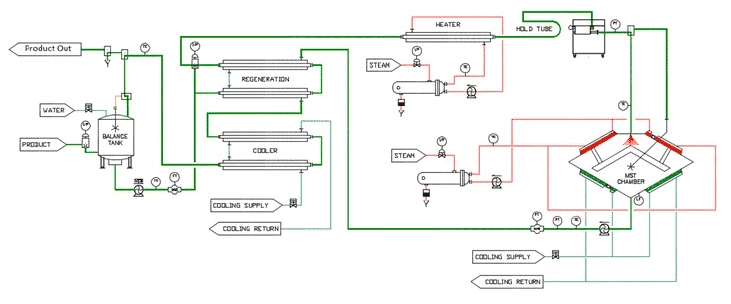
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4899401/#:~:text=The%20LTST%20method%20depends%20on,(%E2%89%A472.7%20%C2%B0C
Incorporation into a Food Safety Management System
Pasteurization is widely used throughout the food industry for its ability to produce a safer product and protect consumers while extending the shelf life of the product and reducing its rate of spoilage.
As such it plays an integral part in many food safety management systems and is a fundamental requirement of many separate pieces of legislation.
In the US, the FDA Grade “A” Pasteurized Milk Ordinance is a minimum requirement that must be followed, although more stringent requirements might be required from individual states.
Legal requirements for milk pasteurization in the EU are set down in:
- Regulation (EC) No 853/2004 (covering hygiene rules for food of animal origin, including pasteurization processing parameters requirements)
- Regulation (EC) No 852/2004 (regulation on food hygiene),
- Regulation (EC) 2073/2005 (microbiological criteria for foodstuffs).
When considering the place of pasteurization for your manufacturing, it is first important to define what the ultimate end goal of the product is desired to be, i.e., is a long shelf life being sought or is it envisioned that the product will be consumed quickly upon consumption?
Once defined, an assessment of pasteurization processes considering the above points and their appropriateness to your manufacturing environment should be considered.
When it comes to including the pasteurization process into your HACCP or Food Safety Plan one could consider beginning with the following steps:
-
- Include pasteurization in the Flow Diagram (and Plant Schematic)
- Ensure there is a clear link to a microbiological hazard and a target pathogen has been identified in the Hazard Analysis
- When applied as a CCP or oPRP include parameters which need to be monitored
- Establish critical limits, corrective actions (also refer to our earlier consideration of a potential Maillard reaction)
- Always include validation and verification for the pasteurization
- Detail cleaning procedures
- Ensure instruments used in calibration are appropriately assessed and monitored
- Create a preventive maintenance plan for the pasteurizer
- Establish a glass and plastic log if relevant
Of course, as with all HAACP and Food Safety Plans, the above considers that you operate an effective documentation and record-keeping system. (Further reading: A Complete Guide to Food Cooking Temperature Control)
We can help with that.
Safefood 360° is a cloud-based Food Safety and Supplier Quality Management System used by dairy and food businesses globally to manage pasteurization and general compliance to all global major and retailer technical standards and legislation.
If you would like to know more information, just click the button below and request a demo to see how the software can assist with your compliance requirements.






I suppose there is incorrect definition for low acid and high acid food, the pH value stated (under section-Purpose) was the other way round,
Hi Sheryl,
You are correct. This was an error in the original document which was corrected. Thanks for spotting it!
Kind regards,
Barry
Nice summary of Pasteurization, thanks.
There seems to be an error though in the discussion about pH. Lower pH means higher acid.
Where it says “In low acidic foods, i.e., those with a pH of less than 4.5….” should instead say “In HIGH acid foods, those with a pH of less that 4.5….”.
And where it says “In foods with a higher acidic value, i.e., those with a pH value of more than 4.6 such as milk….” should instead say “In foods with a LOWER acidic value, i.e., those with a pH value of more than 4.6….”
Regards,
Larry
Hi Larry,
Good spot. This was an error in the original document that has since been corrected.
Kind regards,
Barry
A very useful and interesting article – thank you.
Hi Susan,
Glad you enjoyed the article!
Kind regards,
Barry
Great article, well done and explained. Is it possible to get the article in pdf format?
Hi Hussein,
The article isn’t available as a bespoke pdf, but you are welcome to save the article as the pdf and share it off-line.
Kind regards,
Barry