Understanding the difference between PRP, OPRP & CCP
As a professional in the field of food safety, you’ve likely come across terms such as PRP, CCP and oPRP, and may even find them somewhat perplexing. This comprehensive guide aims to unravel these terminologies’ mysteries and provide clear explanations.
You would be familiar with Prerequisite Program (PRP) and Critical Control Point (CCP), which stems from HACCP (Hazard Analysis and Critical Control Point), These stem from HACCP (Hazard Analysis and Critical Control Point), a strategic risk assessment tool crafted by the Pillsbury Company alongside NASA in the 1960s. This tool aims to pinpoint specific hazards within processes, assess their importance, and devise effective controls to ensure such hazards don’t make it to the consumer – or astronaut, as the case may be.
You might also have encountered the term Operational Prerequisite Program (oPRP), especially if your food safety management aligns with the ISO 22000 standards.
In the following decades since HACCP debuted, many food safety standards, best practice codes, and regulatory guidelines have adapted and adopted the HACCP principles (Where now for HACCP?). They’ve brought forth their interpretations of how these principles should be applied along the way.
The purpose of risk assessment and management
When we speak of HACCP, risk assessment and determination of control measures is an aspect which many people may find difficult, if not mystifying, on occasion.
Simply put, the basic principle is to install control measures appropriate for the hazards and risks they pose to the final consumer within the process and operation.
HACCP requires you to identify these potential hazards and then determine the significance of these hazards by applying risk assessment techniques, the output of which is a measure of risk which then allows us to put in place appropriate control measures, such as PRP, oPRP, and CCP.
Over the decades, various standards have attempted to define how risk assessment is carried out and how to determine control measures.
In the process, they have introduced their unique terms, language, scope, methodology and workflows. While some of these have been good, much of this work has served only to confuse people causing HACCP plans which are unnecessarily complex and hinder the effective management of food safety.
Various standards have attempted to define how risk assessment is carried out and how to determine control measures…only to confuse people causing HACCP plans which are unnecessarily complex and hinder the effective management of food safety
In the context of food safety, the International Standards Organisation (ISO) has defined a control measure as an action or activity that can be used to prevent or eliminate a food safety hazard or reduce it to an acceptable level.
This definition is general and can be used to describe virtually any action, step, activity, job, task, process or procedure intending to address a food safety hazard. Consider downloading our most recent datasheet on food safety hazards for additional information.
As we look closer at the main food safety standards, we can see that control measures have become categorized.
Categories of Control Measures
Prerequisite Program (PRP)
ISO defines the basic conditions and activities necessary to maintain a hygienic environment throughout the food chain suitable for the production, handling and provision of safe end products and safe food for human consumption.
A wide variety of PRPs depend on the particular product and process.
They are often described in certain sectors of the industry as Good Agricultural Practice (GAP), Good Veterinarian Practice (GVP), Good Manufacturing Practice (GMP), Good Hygienic Practice (GHP), Good Production Practice (GPP), Good Distribution Practice (GDP), and Good Trading Practice (GTP).
Examples of PRPs in a food manufacturing environment would be:
- construction and layout of buildings and associated utilities
- the layout of premises, including workspace and employee facilities
- supplies of air, water, energy and other utilities
- supporting services, including waste and sewage disposal
- the suitability of the equipment and its accessibility for cleaning, maintenance and preventative maintenance
- management of purchased materials (e.g., raw materials, ingredients, chemicals and packaging), supplies (e.g., water, air, steam and ice), disposals (e.g., waste and sewage) and handling of products (e.g., storage and transportation)
- measures for the prevention of cross-contamination
- cleaning and sanitizing
- pest control
- personnel hygiene
PRPs are usually general to the process and not focused on any particular step in the process.
For example, cleaning and sanitizing are activities which can apply to all steps, rooms, items and building fabric.
They also have the character that their failure does not necessarily lead to an immediate and imminent food safety risk.
Failure of a PRP does not necessarily lead to an immediate and imminent food safety risk
PRPs have the characteristic that their failure does not instantaneously trigger a direct food safety risk. The failure of a PRP does not immediately pose a serious threat to food safety. Typically, a prolonged period and repeated failures are necessary to shift product safety significantly.
In simpler terms, PRPs are typically designed to manage more general and lower-risk hazards within the food production and distribution process.
Critical Control Point (CCP)
The Critical Control Point (CCP), one of the most well-known components in food safety control measures, is defined by the ISO as a specific step in which control must be implemented. It’s crucial to prevent, eliminate, or reduce a food safety hazard to an acceptable level.
This definition is similar to the general definition of a control measure. However, a CCP differs as it relates specifically to a step in the process, such as cooking, cooling, or freezing, and not a general activity or action.
a CCP differs from a control measure as it relates specifically to a step in the process, and not a general activity or action
ISO also states that a CCP is a step at which control can be applied, which means that if a ‘CCP’ cannot apply control, it cannot be considered a CCP.
In scenarios where control is subjective and cannot be measured accurately, this may be problematic as control cannot be enforced.
If a CCP is unable to apply control, it cannot be considered a CCP
Furthermore, the risk that a hazard poses should the control not be executed is another crucial aspect related to CCPs. For example, if Salmonella contaminates cooked meat, failing to cook the meat at the proper temperature and for the necessary duration exposes consumers to significant health risks. In such scenarios, the control step becomes critical, specifically designed and strictly enforced to manage the hazard.
Operational Prerequisite Program (OPRP)
The concept of the Operational Prerequisite Program (oPRP) was introduced by the ISO in their food safety management standard, ISO 22000. It is defined as a control measure that the hazard analysis identifies as critical to control the likelihood of introducing food safety hazards or the contamination or proliferation of these hazards in the product(s) or the processing environment.
What is striking about this control measure and its definition is the almost universal confusion users have in differentiating it from PRP and CCP.
To shed light on what an oPRP truly is, one can think of it as a particular action linked to the process. Although it may not be critical for food safety, it plays an indispensable role in mitigating the likelihood of a specific hazard’s occurrence.
oPRP’s are essential in reducing the likelihood of a specific hazard occurring
Let’s take the example of a cooking step in a food preparation process. This step may be crucial for controlling the risk of a specific pathogen, such as E. coli, surviving. It forms an inherent part of producing a cooked product, and removing it from the process isn’t feasible. Therefore, the control applied at this step can be considered critical, and as such, it is categorized as a CCP.
Now, consider a metal detection process in the same food production line. Like the cooking step, it aims to reduce the chances of a hazard reaching the consumer, which could arguably categorize it as a CCP as well. However, the crucial difference lies in the fact that metal detection is not an inherent step required to produce safely cooked food. The process can function without it, and the company can still produce relatively safe products. Nonetheless, the presence of metal detection could be deemed essential to reduce the likelihood of a hazard, hence qualifying it as an oPRP.
It is important to realize that this is simply one possible definition of an oPRP which provides some working understanding.
Comparison Table: PRP vs. CCP vs. OPRP – Differences in Food Safety
| PRP (Prerequisite Program) | CCP (Critical Control Point) | oPRP (Operational Prerequisite Program) | |
|---|---|---|---|
| Definition | Basic conditions and activities necessary to maintain a hygienic environment throughout the food chain, suitable for the production of safe food. | A step in a food process at which control can be applied, and a food safety hazard can be prevented, eliminated, or reduced to acceptable levels. | A control measure that the hazard analysis identifies as necessary to control the likelihood of introducing food safety hazards or the contamination or proliferation of these hazards in the product(s) or the processing environment. |
| Purpose | Ensure a clean and safe environment for food production. | Specifically control identified hazards critical to food safety. | Control the likelihood of food safety hazards. Not necessarily critical, but essential in reducing the risk of specific hazards. |
| Example | Pest control, personal hygiene, cleaning and sanitation, training. | Cooking, cooling, reheating, specific cleaning procedures. | Steps like metal detection, sieving, specific temperature or humidity control in storage. |
| Identification in the industry | Determined by good manufacturing practices and do not usually require monitoring. | Identified through a HACCP study, typically require critical limits, monitoring, corrective action, verification, and record keeping. | Identified by the hazard analysis, often require validation, monitoring, corrective action, and record keeping, but usually do not have critical limits. |
When to determine and apply control points
GFSI Standards set out requirements for determining control points. The requirements under BRC v8 and ISO 22000:2018 do vary, so it’s important to understand each fully and how they apply to your specific situation.
| 2.8 | DETERMINE THE CRITICAL CONTROL POINTS (CCPS) (EQUIVALENT TO CODEX ALIMENTARIUS STEP 7, PRINCIPLE 2) |
|---|---|
| CLAUSE | REQUIREMENTS |
| 2.8.1 | For each hazard that requires control, control points shall be reviewed to identify those that are critical. This requires a logical approach and may be facilitated by use of a decision tree. Critical control points (CCPs) shall be those control points which are required in order to prevent or eliminate a food safety hazard or reduce it to an acceptable level. If a hazard is identified at a step where control is necessary for safety but the control does not exist, the product or process shall be modified at that step, or at an earlier step, to provide a control measure. |
| 8.5.2.4 | Selection and categorization of control measure(s) |
|---|---|
| 8.5.2.4.1
8.5.2.4.2 |
Based on the hazard assessment, the organization shall select an appropriate control measure or combination of control measures that will be capable of preventing or reducing the identified significant food safety hazards to defined acceptable levels.
The organization shall categorize the selected identified control measure(s) to be managed as OPRP(s) (see 3.30) or at CCPs (see 3.11). The categorization shall be carried out using a systematic approach. For each of the control measures selected, there shall be an assessment of the following: a) the likelihood of failure of its functioning; b) the severity of the consequence in the case of failure of its functioning; this assessment shall include:
In addition, for each control measure, the systematic approach shall include an assessment of the feasibility of: a) establishing measurable critical limits and/or measurable/observable action criteria; b) monitoring to detect any failure to remain within critical limit and/or measurable/observable action criteria; c) applying timely corrections in case of failure. The decision-making process and results of the selection and categorization of the control measures shall be maintained as documented information. External requirements (e.g. statutory, regulatory and customer requirements) that can impact the choice and the strictness of the control measures shall also be maintained as documented information. |
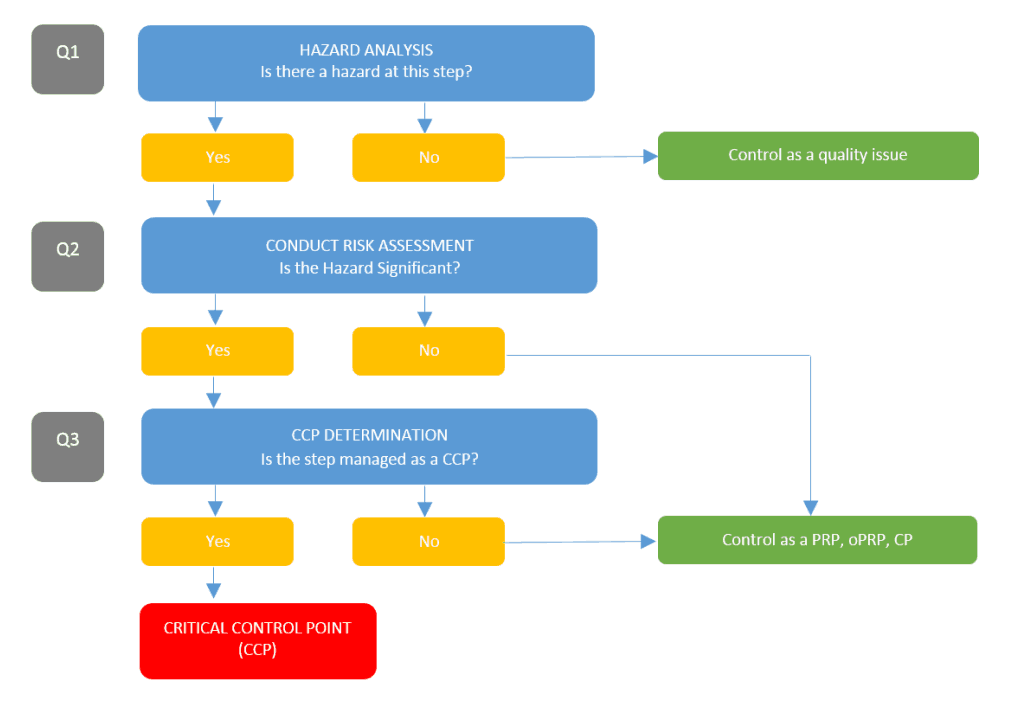
After specific hazards are identified at a process step, a risk assessment is required to determine if the hazard is significant or not.
If significant hazards are identified then a decision tree is required to help determine if the hazard requires control and, if so, should they be controlled as a CCP, PRP or oPRP.
Significance describes those hazards which present a real risk of impacting on the consumer.
It may be said that significance is essentially an expression of risk.
In food safety, risk is a measure of the combined severity of impact from a hazard and its probability of occurrence.
risk is a measure of the combined severity of impact from a hazard and its probability of occurrence
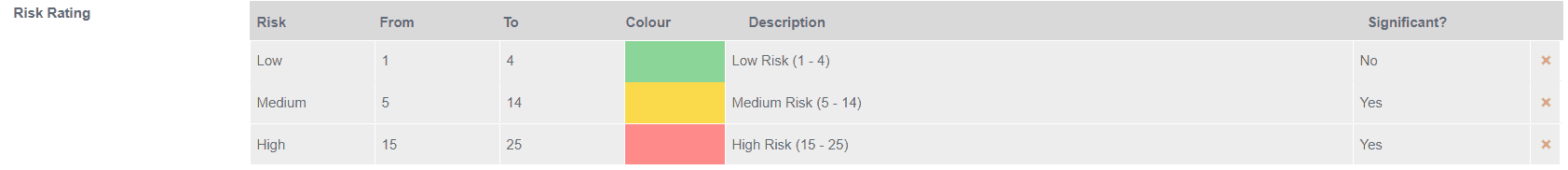
In its simplest form risk is expressed as being High, Medium or Low.
Below is an example of a 5 x 5 risk assessment model along with a risk rating which uses Safefood 360° to demonstrate.
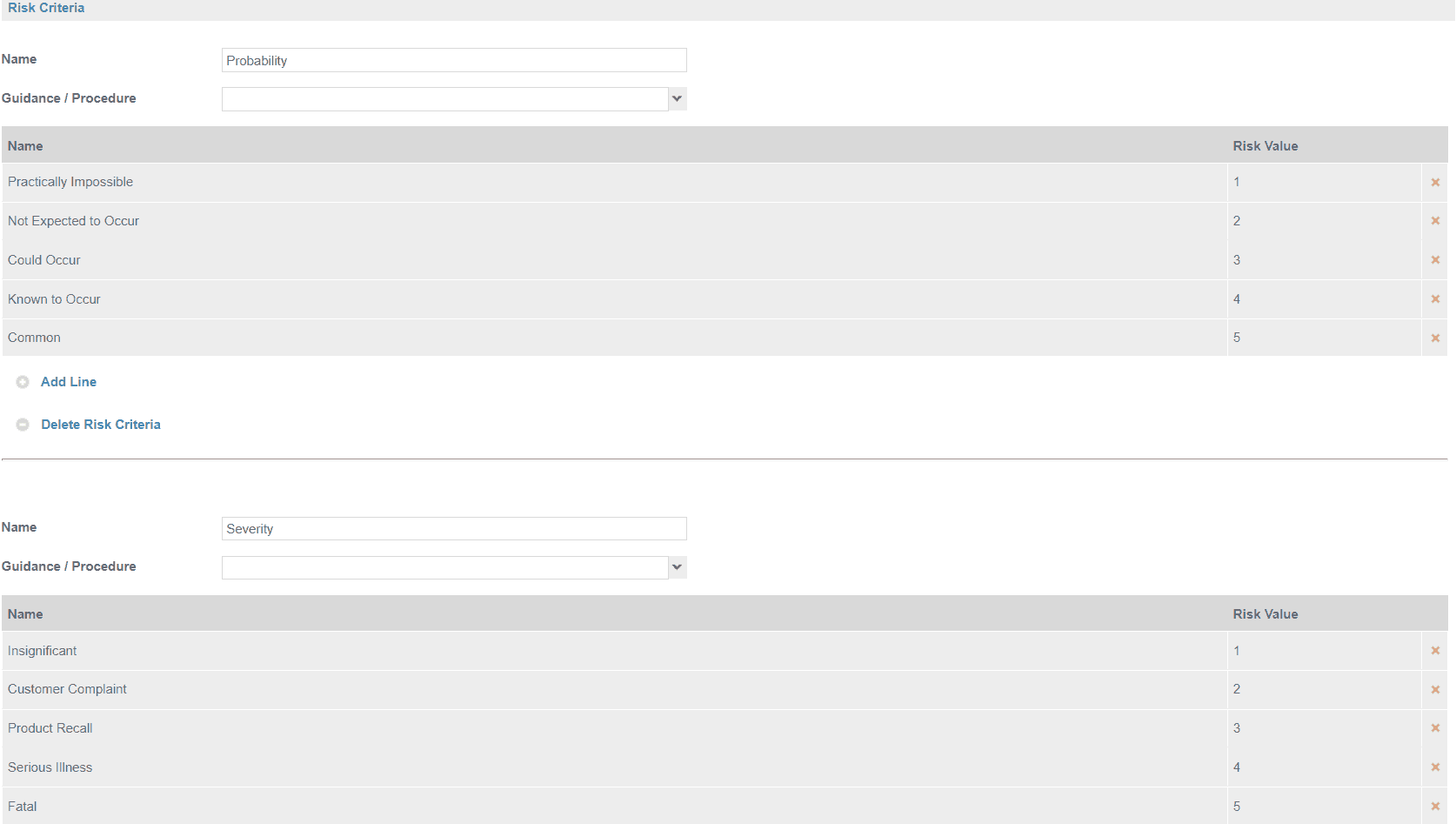
If significant hazards are identified then a decision tree is required to help determine if the hazard requires control and if so should they be controlled as a CCP, PRP or oPRP.
There are large numbers of decision trees employed and cited in various standards, but arguably the most common decision tree is the CODEX standard decision tree for HACCP.
The CODEX tree focuses on whether the hazard should be controlled as a CCP or not, but it’s important to be aware that it doesn’t assist in determining what type of control shall be used where there is an outcome other than CCP.
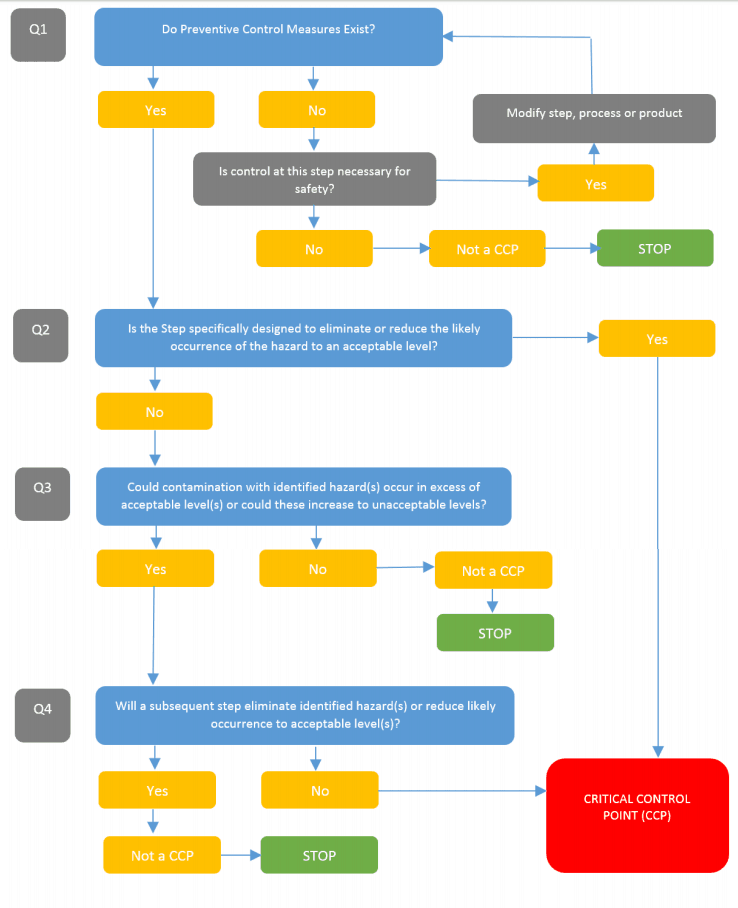
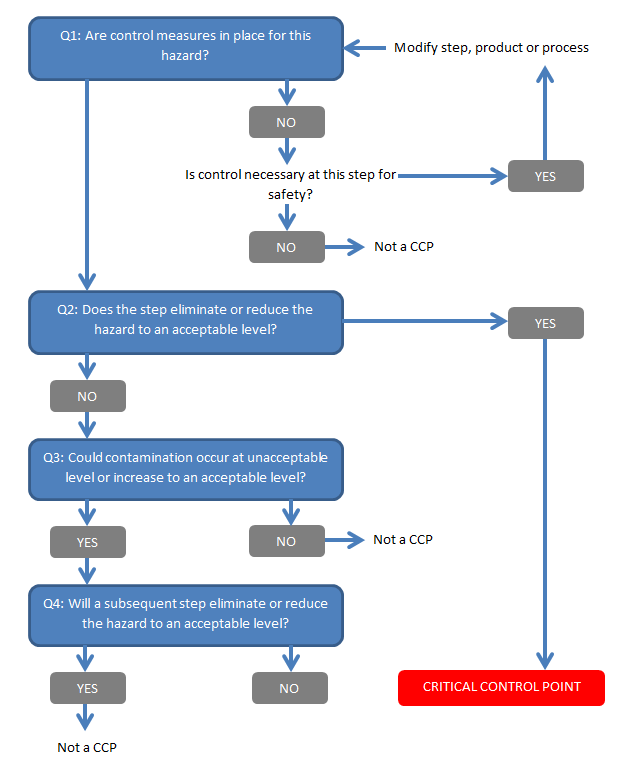
Other robust models can account for oPRP and PRP.
Simple Decision Tree
CODEX Decision Tree
Robust Example
The more robust model really builds on the CODEX decision tree.
The tree allows for a solid and logical approach to determine control measures and will clearly show an auditor how you arrived at your decisions.
Risk assessment models and decision trees will help you to determine hazard management and controls.
It is clear that PRP’s are basic controls/activities within a production facility.
oPRP’s and CCP’s are specific and can be determined as being the control by using a decision tree.
Applying these steps in the future
Understanding the application and definition of control points within the framework you comply with is crucial when determining their relevance and applicability. This is particularly important as it can vary under GFSI (Global Food Safety Initiative) standards. It is essential to read and comprehend these rules thoroughly and how they pertain to your specific situation to effectively apply the information and your understanding of control.
Implementing an effective control process is a significant step towards mitigating risks and ensuring safe and high-quality food production for your customers. While various other steps and approaches can be combined to enhance effectiveness, Safefood 360° offers a robust system that assists in applying control points within the context of your overall food safety management system.
If you would like more information on how you can benefit from Safefood 360° and its capabilities, please feel free to consult with our technical experts by contacting our technical consultant here.